- +1
出版简讯|健康血浆治疗通过FGF21保护卒中小鼠血脑屏障,并改善卒中预后
编者荐语:
来源:“SVN俱乐部”微信公众号,扫描文末二维码,更多学术前沿热点资讯即将上线,敬请关注!
以下文章来源于SVN俱乐部 ,作者汤耀辉,杨国源
 SVN俱乐部
SVN俱乐部由中国卒中学会官方英文期刊 Stroke and Vascular Neurology(SVN)编辑部创办,向广大卒中领域同仁推送最新出版简讯、SVN中文解读、期刊投审稿指南、国际卒中领域相关出版资讯等
 Plasma from healthy donors protects blood–brain barrier integrity via FGF21 and improves the recovery in a mouse model of cerebral ischaemia
Plasma from healthy donors protects blood–brain barrier integrity via FGF21 and improves the recovery in a mouse model of cerebral ischaemiaMuyassar Mamtilahun, Lu Jiang, Yaying Song, Xiaojing Shi, Chang Liu, Yixu Jiang, Lidong Deng, Haoran Zheng, Hui Shen, Yongfang Li, Zhijun Zhang, Yongting Wang, Yaohui Tang, Guo-Yuan Yang
doi: 10.1136/svn-2020-000774
 Stroke & Vascular Neurology(SVN)最新上线文章“Plasma from healthy donors protects blood–brain barrier integrity via FGF21 and improves the recovery in a mouse model of cerebral ischaemia”,来自上海交通大学生物医学工程学院Med-X研究院杨国源教授团队,并获得中国科技部国家重点研发计划“重大慢性非传染性疾病防控研究”专项、上海市教委创新基金、国家自然科学基金等支持。
Stroke & Vascular Neurology(SVN)最新上线文章“Plasma from healthy donors protects blood–brain barrier integrity via FGF21 and improves the recovery in a mouse model of cerebral ischaemia”,来自上海交通大学生物医学工程学院Med-X研究院杨国源教授团队,并获得中国科技部国家重点研发计划“重大慢性非传染性疾病防控研究”专项、上海市教委创新基金、国家自然科学基金等支持。健康血浆治疗可逆转认知缺陷,并促进衰老相关脑疾病的神经可塑性。然而,健康血浆治疗能否改善卒中后血脑屏障完整性尚不清楚。本研究中,作者团队应用健康雌性小鼠血浆连续8次尾静脉注射治疗(90分钟)短暂性大脑中动脉栓塞(tMCAO)的成年雌性小鼠。通过检测梗塞体积、脑萎缩和神经运动功能来评估血浆治疗对缺血性卒中小鼠预后的影响。并通过细胞凋亡、血脑屏障完整性和成纤维细胞生长因子21(Fibroblast growth factor21, FGF21)基因敲除小鼠来研究其潜在的作用机制。
本研究发现,健康血浆注射可促进卒中后神经运动功能恢复,并减少梗死体积、脑水肿和脑萎缩。免疫荧光染色结果显示,与对照组相比,血浆注射卒中小鼠脑组织梗死边缘区中凋亡的神经元细胞数量减少。与此同时,健康血浆注射减少缺血性卒中后3天时血脑屏障紧密连接蛋白的降解以及IgG的渗漏。免疫印迹结果表明,健康血浆注射显著增加了卒中小鼠脑组织中FGF21蛋白表达。而当把FGF21基因敲除小鼠的血浆注射到野生型卒中小鼠后,并没有看到健康血浆对血脑屏障的保护作用,表明健康血浆主要是通过FGF21保护小鼠的血脑屏障。
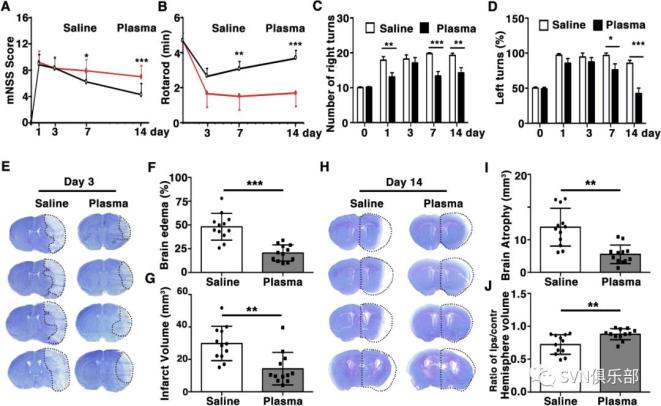 Figure 1. Healthy plasma treatment promoted neurobehavioural recovery, reduced infarct volume and atrophy volume in tMCAO mice bar graphs showed that the results of modified neurological severity score (A), rotarod test (B), EBST (C) and corner test (D) in plasma-injected and control mice at 1, 3, 7 and 14 days after tMCAO. Representative images of cresyl violet-stained brain sections of plasma-injected and control mice at 3 days (E) and 14 days (H) after tMCAO. Bar graph showed quantification of brain oedema and infarct volume in plasma-injected and control mice at 3 days (F, G) and brain atrophy volume at 14 days (I, J) after tMCAO. Data are mean±SD, n=12 per group. *p<0.05, **p<0.01. ***p<0.001, plasma-injected versus control mice. EBST, elevated body swing test; tMACO, transient middle cerebral artery occlusion.
Figure 1. Healthy plasma treatment promoted neurobehavioural recovery, reduced infarct volume and atrophy volume in tMCAO mice bar graphs showed that the results of modified neurological severity score (A), rotarod test (B), EBST (C) and corner test (D) in plasma-injected and control mice at 1, 3, 7 and 14 days after tMCAO. Representative images of cresyl violet-stained brain sections of plasma-injected and control mice at 3 days (E) and 14 days (H) after tMCAO. Bar graph showed quantification of brain oedema and infarct volume in plasma-injected and control mice at 3 days (F, G) and brain atrophy volume at 14 days (I, J) after tMCAO. Data are mean±SD, n=12 per group. *p<0.05, **p<0.01. ***p<0.001, plasma-injected versus control mice. EBST, elevated body swing test; tMACO, transient middle cerebral artery occlusion.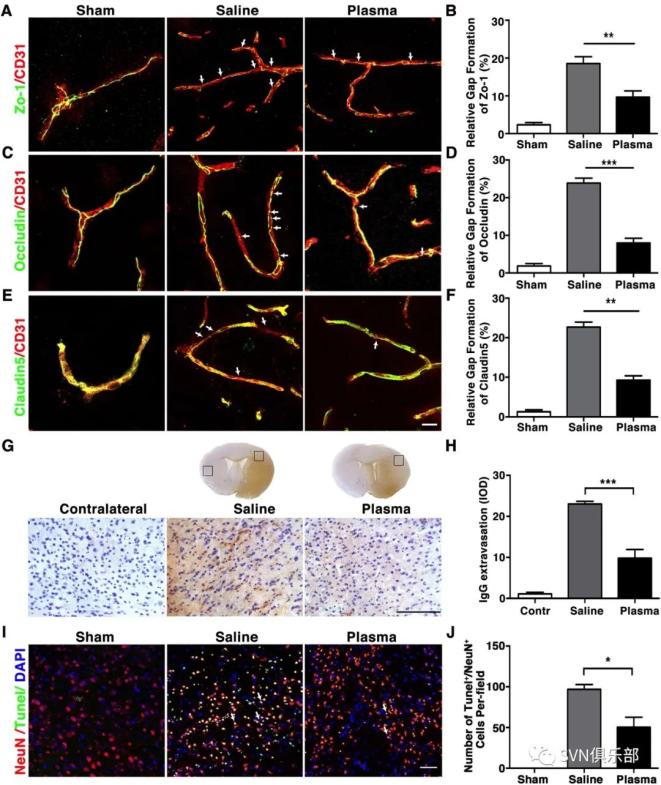 Figure 2. Healthy plasma reduced BBB disruption and neuronal apoptosis in tMCAO mice photomicrographs showed that the endothelial cell marker CD31 (red) with BBB tight junction proteins (green), ZO-1 (A), occludin (C) and claudin-5 (E) in the peri-infarct area of the striatum in sham, salineinjected and plasma-injected mice. Arrows indicate that the gaps in BBB tight junctions. Scale bar=10 µm. Bar graphs showed that quantification of BBB tight junction proteins ZO-1 (B), occludin (D) and claudin-5 (E) in sham, saline-injected and plasma-injected mice at 3 days of tMCAO. Data are mean±SD, n=4 per group. **p<0.01. ***p<0.001, Plasma-injected versus control mice. (G) Photomicrographs showed that the IgG (brown) in the peri-infarct area of the striatum in sham, saline-injected and plasma-injected mice. Scale bar=75 μm. Bar graphs showed the quantification of IgG proteins (H) in sham, saline-injected and plasma-injected mice at 3 days of tMCAO. Data are mean±SD, n=5 per group. ***p<0.001, plasma injected versus control mice. (I) Photomicrographs showed that the NeuN (red) and TUNEL (green) doublepositive cells in the peri-focal area in the plasma-treated and control mice at 3 days of tMCAO. Arrows indicate NeuN+/TUNEL+ cells, scale bar=50 μm. (J) Bar graphs showed the quantification of TUNEL+ cells in plasma-treated sham, and control mice at 3 days of tMCAO. Data are mean±SD, n=5 per group. * p<0.05, plasma injected versus saline injected. BBB, blood–brain barrier; tMCAOM, transient middle cerebral artery occlusion; TUNEL, transferase dUTP nick end labelling; ZO-1, zonula occludens-1.
Figure 2. Healthy plasma reduced BBB disruption and neuronal apoptosis in tMCAO mice photomicrographs showed that the endothelial cell marker CD31 (red) with BBB tight junction proteins (green), ZO-1 (A), occludin (C) and claudin-5 (E) in the peri-infarct area of the striatum in sham, salineinjected and plasma-injected mice. Arrows indicate that the gaps in BBB tight junctions. Scale bar=10 µm. Bar graphs showed that quantification of BBB tight junction proteins ZO-1 (B), occludin (D) and claudin-5 (E) in sham, saline-injected and plasma-injected mice at 3 days of tMCAO. Data are mean±SD, n=4 per group. **p<0.01. ***p<0.001, Plasma-injected versus control mice. (G) Photomicrographs showed that the IgG (brown) in the peri-infarct area of the striatum in sham, saline-injected and plasma-injected mice. Scale bar=75 μm. Bar graphs showed the quantification of IgG proteins (H) in sham, saline-injected and plasma-injected mice at 3 days of tMCAO. Data are mean±SD, n=5 per group. ***p<0.001, plasma injected versus control mice. (I) Photomicrographs showed that the NeuN (red) and TUNEL (green) doublepositive cells in the peri-focal area in the plasma-treated and control mice at 3 days of tMCAO. Arrows indicate NeuN+/TUNEL+ cells, scale bar=50 μm. (J) Bar graphs showed the quantification of TUNEL+ cells in plasma-treated sham, and control mice at 3 days of tMCAO. Data are mean±SD, n=5 per group. * p<0.05, plasma injected versus saline injected. BBB, blood–brain barrier; tMCAOM, transient middle cerebral artery occlusion; TUNEL, transferase dUTP nick end labelling; ZO-1, zonula occludens-1.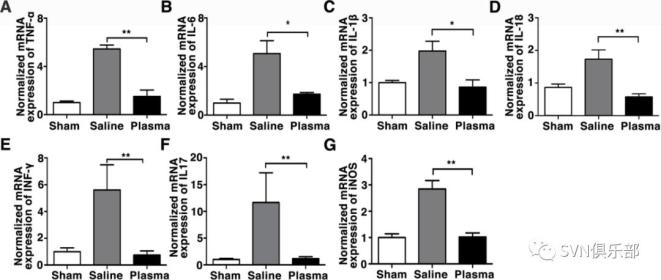 Figure 3. Healthy plasma reduced inflammatory response in the tMCAO mice brain bar graph showed that RT-PCR quantification of inflammatory factors TNF-α (A), IL-6 (B), IL-1β (C), IL-18 (D), INF-γ (E), IL-17 (F) and iNOS (G) mRNA expression in sham, saline-injected and plasma-injected mouse brain at 3 days of tMCAO. data are mean±SD, n=6 per group. *p<0.05; **p<0.01, plasma injected versus control mice. INF-γ, interferon-γ; IL-6, interleukin 6; iNOS, inducible nitric oxide synthase; tMCAO, transient middle cerebral artery occlusion; TNF-α, tumour necrosis factor-α.
Figure 3. Healthy plasma reduced inflammatory response in the tMCAO mice brain bar graph showed that RT-PCR quantification of inflammatory factors TNF-α (A), IL-6 (B), IL-1β (C), IL-18 (D), INF-γ (E), IL-17 (F) and iNOS (G) mRNA expression in sham, saline-injected and plasma-injected mouse brain at 3 days of tMCAO. data are mean±SD, n=6 per group. *p<0.05; **p<0.01, plasma injected versus control mice. INF-γ, interferon-γ; IL-6, interleukin 6; iNOS, inducible nitric oxide synthase; tMCAO, transient middle cerebral artery occlusion; TNF-α, tumour necrosis factor-α.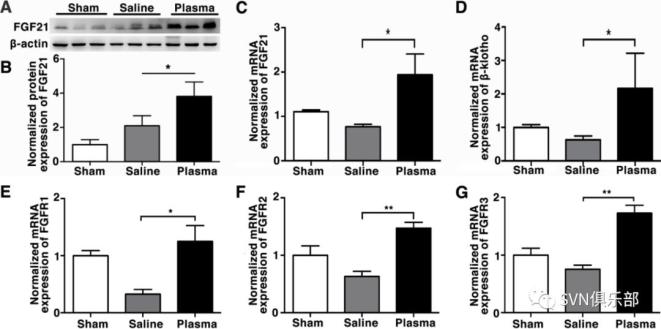 Figure 4. Healthy plasma increased FGF21 and its' receptors expression in the tMCAO mouse brain photomicrograph showed that Western blot images of FGF21 bands (A) and bar graph (B) showed semiquantification of FGF21 protein expression in sham, saline-injected and plasma-injected mouse brain at 3 days of tMCAO. Data are mean±SD, n=3 per group. *p<0.05, plasma-injected versus control mice. RT-PCR quantification of FGF21 (C), β-klotho (D), FGFR1 (E), FGFR2 (F) and FGFR3 (G) mRNA expression in the sham, saline-injected and plasma-injected mouse brain at 3 days of tMCAO. Data are mean±SD, n=4 per group. *p<0.05; ** p<0.01, plasma-injected versus control. FGF21, fibroblast growth factor 21; FGFR1, fibroblast growth factor receptor; tMCAO, transient middle cerebral artery occlusion.
Figure 4. Healthy plasma increased FGF21 and its' receptors expression in the tMCAO mouse brain photomicrograph showed that Western blot images of FGF21 bands (A) and bar graph (B) showed semiquantification of FGF21 protein expression in sham, saline-injected and plasma-injected mouse brain at 3 days of tMCAO. Data are mean±SD, n=3 per group. *p<0.05, plasma-injected versus control mice. RT-PCR quantification of FGF21 (C), β-klotho (D), FGFR1 (E), FGFR2 (F) and FGFR3 (G) mRNA expression in the sham, saline-injected and plasma-injected mouse brain at 3 days of tMCAO. Data are mean±SD, n=4 per group. *p<0.05; ** p<0.01, plasma-injected versus control. FGF21, fibroblast growth factor 21; FGFR1, fibroblast growth factor receptor; tMCAO, transient middle cerebral artery occlusion.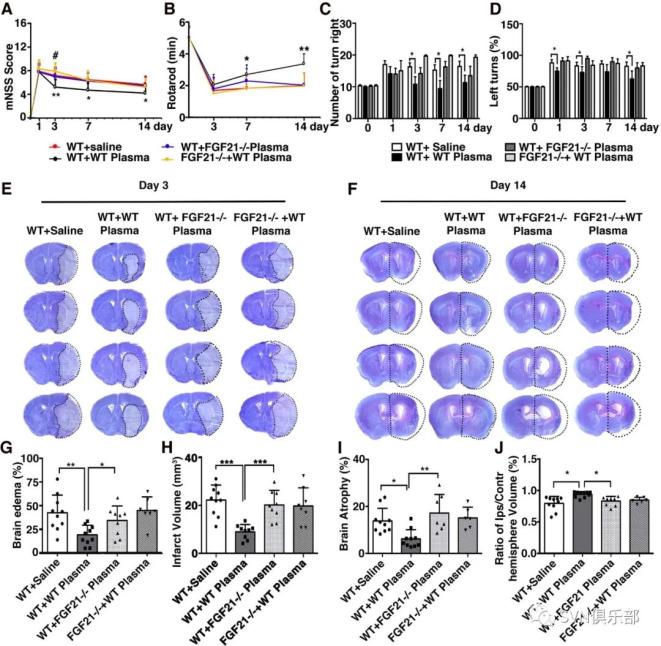 Figure 5. Depletion of FGF21 in plasma reversed the beneficial effect of healthy plasma in the tMCAO mice bar graphs showed that neurological severity score (A), rotarod test (B), EBST (C) and corner test (D) in wild-type mice that treated with plasma from wild-type mice (WT+WT); wild-type mice that treated with plasma from FGF21-/- mice (WT+ FGF21-/- plasma); and FGF21-/- mice that treated with wild-type plasma (FGF21-/-+ WT plasma) at 1, 3, 7 and 14 days after tMCAO. (E, F) Images of cresyl violet-stained brain sections of WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group, and FGF21-/-+ WT plasma group at 3 and 14 days of tMCAO. Dashed line showed infarct area in the ipsilateral hemisphere of the brain. Semiquantification of brain oedema and infarct volume in WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group, and FGF21-/-+ WT plasma group at 3 days of tMCAO (G, H). Bbar graph showed that the brain atrophy volume and the volume ratio of the ipsilateral hemisphere and contralateral hemisphere in the WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group and FGF21-/-+ WT plasma group at 14 days in the tMCAO mouse brain (I, J). Data are mean±SD, n=10 per group. *p<0.05, **p<0.01, ***p<0.001, FGF21-treated versus control mice. EBST, elevated body swing test; FGF21, fibroblast growth factor 21; tMCAO, transient middle cerebral artery occlusion.
Figure 5. Depletion of FGF21 in plasma reversed the beneficial effect of healthy plasma in the tMCAO mice bar graphs showed that neurological severity score (A), rotarod test (B), EBST (C) and corner test (D) in wild-type mice that treated with plasma from wild-type mice (WT+WT); wild-type mice that treated with plasma from FGF21-/- mice (WT+ FGF21-/- plasma); and FGF21-/- mice that treated with wild-type plasma (FGF21-/-+ WT plasma) at 1, 3, 7 and 14 days after tMCAO. (E, F) Images of cresyl violet-stained brain sections of WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group, and FGF21-/-+ WT plasma group at 3 and 14 days of tMCAO. Dashed line showed infarct area in the ipsilateral hemisphere of the brain. Semiquantification of brain oedema and infarct volume in WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group, and FGF21-/-+ WT plasma group at 3 days of tMCAO (G, H). Bbar graph showed that the brain atrophy volume and the volume ratio of the ipsilateral hemisphere and contralateral hemisphere in the WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group and FGF21-/-+ WT plasma group at 14 days in the tMCAO mouse brain (I, J). Data are mean±SD, n=10 per group. *p<0.05, **p<0.01, ***p<0.001, FGF21-treated versus control mice. EBST, elevated body swing test; FGF21, fibroblast growth factor 21; tMCAO, transient middle cerebral artery occlusion.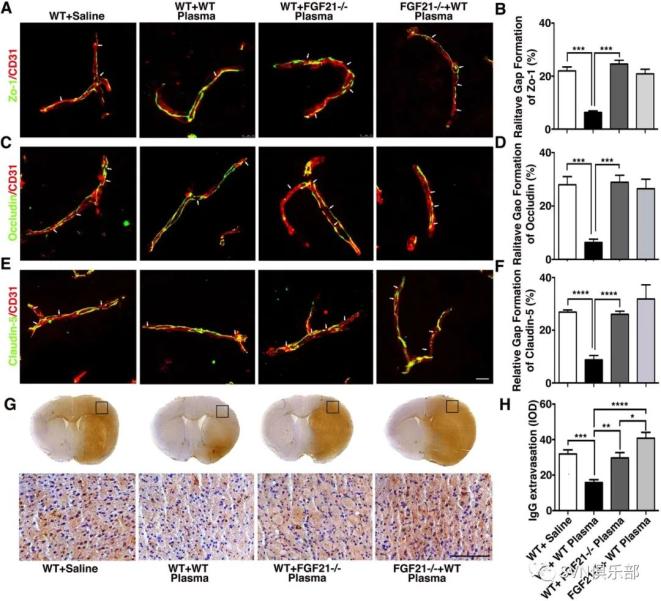 Figure 6. Depletion of FGF21 in plasma diminished the beneficial effects of plasma-induced BBB protection in mice after 3 days of tMCAO coimmunostaining of endothelial cell marker CD31(red) with BBB (green) ZO-1(A), occludin (C) and claudin-5 (E) in the peri-infarct area of the striatum in wild-type mice that treated with saline (WT+ saline), wild-type mice that treated with wild mouse plasma (WT+WT), wild-type mice that treated with plasma from FGF21-/- mice (WT+ FGF21-/- plasma) and FGF21-/- KO mice that treated with wild-type plasma (FGF21-/- + WT plasma) at 3 days of tMCAO. Arrows indicate the gaps in BBB tight junction proteins, scale bar=10 μm. Quantification of gaps that formed on BBB tight junction proteins ZO-1(B), occludin (D) and claudin-5 (F) in the WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group, and FGF21-/-+ WT plasma group at 3 days of tMCAO. Data are mean±SD, n=3 per group. **p<0.01. ***p<0.001. (G) Photomicrographs of IgG in the perifocal area of WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group and FGF21-/-+ WT plasma group at 3 days after stroke. Scale bar=75 μm. (H) Bar graph showed semiquantification of IgG extravasation in the WT+ Saline group, WT+WT group, WT+FGF21-/- plasma group and FGF21-/-+WT plasma group. Data are presented as mean±SD, n=5 per group. * p<0.05; ****p<0.001, plasma versus control. BBB, blood-brain barrier; FGF21, fibroblast growth factor 21; tMCAO, transient middle cerebral artery occlusion; ZO-1, zonula occludens-1.
Figure 6. Depletion of FGF21 in plasma diminished the beneficial effects of plasma-induced BBB protection in mice after 3 days of tMCAO coimmunostaining of endothelial cell marker CD31(red) with BBB (green) ZO-1(A), occludin (C) and claudin-5 (E) in the peri-infarct area of the striatum in wild-type mice that treated with saline (WT+ saline), wild-type mice that treated with wild mouse plasma (WT+WT), wild-type mice that treated with plasma from FGF21-/- mice (WT+ FGF21-/- plasma) and FGF21-/- KO mice that treated with wild-type plasma (FGF21-/- + WT plasma) at 3 days of tMCAO. Arrows indicate the gaps in BBB tight junction proteins, scale bar=10 μm. Quantification of gaps that formed on BBB tight junction proteins ZO-1(B), occludin (D) and claudin-5 (F) in the WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group, and FGF21-/-+ WT plasma group at 3 days of tMCAO. Data are mean±SD, n=3 per group. **p<0.01. ***p<0.001. (G) Photomicrographs of IgG in the perifocal area of WT+ saline group, WT+WT group, WT+ FGF21-/- plasma group and FGF21-/-+ WT plasma group at 3 days after stroke. Scale bar=75 μm. (H) Bar graph showed semiquantification of IgG extravasation in the WT+ Saline group, WT+WT group, WT+FGF21-/- plasma group and FGF21-/-+WT plasma group. Data are presented as mean±SD, n=5 per group. * p<0.05; ****p<0.001, plasma versus control. BBB, blood-brain barrier; FGF21, fibroblast growth factor 21; tMCAO, transient middle cerebral artery occlusion; ZO-1, zonula occludens-1.研究结果表明,健康血浆治疗通过FGF21促进神经运动功能恢复,保护卒中后血脑屏障的结构和功能完整性,减少神经元凋亡,为缺血性卒中的治疗开辟了新途径。
SVN编辑部(中国)
官方网站:www.svn-csajournal.org (or svn.bmj.com)
Email:csa.svn@chinastroke.net
地址:首都医科大学附属北京天坛医院国家神经系统疾病临床医学研究中心504室
Tel:010-59975024
SVN,期待您的支持与关注!
原标题:《出版简讯|健康血浆治疗通过FGF21保护卒中小鼠血脑屏障,并改善卒中预后》
本文为澎湃号作者或机构在澎湃新闻上传并发布,仅代表该作者或机构观点,不代表澎湃新闻的观点或立场,澎湃新闻仅提供信息发布平台。申请澎湃号请用电脑访问http://renzheng.thepaper.cn。





- 报料热线: 021-962866
- 报料邮箱: news@thepaper.cn
互联网新闻信息服务许可证:31120170006
增值电信业务经营许可证:沪B2-2017116
© 2014-2026 上海东方报业有限公司




